 
So if this ratio was 3:1 that means there are 3 particles of 35Cl for every particle of 37Cl, and the percent abundance would be 75% 35Cl and 25% 37Cl.įigure 2.3. Note, the mass spectrum in figure 2.3.2 (b) gives the relative abundance of each isotope, with the peak normalized to the isotope with the highest abundance. The angle of deflection depends on both the mass of the particle and the magnetic field strength, with the lighter particles being deflected more ( the lighter 35Cl + ions are deflected more than the heavier 37Cl + ions.) At the end of the chamber is an exit hole with a detector, and as the magnetic field intensity is increased the deflection angle changes, which separates the particles. The metal forms a dark oxide layer when it is exposed to air. An alkaline earth metal, strontium is a soft silver-white yellowish metallic element that is highly chemically reactive. These are then accelerated down the chamber until they reach a magnetic field that deflects the particles. Strontium is the chemical element with the symbol Sr and atomic number 38. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of strontium-88 (atomic number. The chlorine has multiple isotopes and is hit with a stream of ionizing electrons which break the bond of Cl 2 and strips electrons off the chlorine causing ions to form. 
In figure 2.3.2 you can see chlorine gas entering an mass spectrometer. mass number (mass : charge ratio).Īlthough we cannot directly measure the mass of atoms, we can use Mass Spectrometer, an instrument that allows us to measure the mass to charge ratio. It was used to make much of the SR-71 Blackbird, the worlds fastest manned aircraft, as well as a major parts of the engines and airframe of the big passenger aircraft including 747s. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Strontium, symbol Sr, has a Face Centered Cubic structure and Silver. How do we know what the percent abundance for each of the isotopes of a given element? Isotopes are separated through mass spectrometry MS traces show the relative abundance of isotopes vs. Element Titanium (Ti), Group 4, Atomic Number 22, d-block, Mass 47.867. Element 38 of Periodic table is Strontium with atomic number 38, atomic weight 87.62. Here is an interesting IUPAC technical report, "Isotope-Abundance Variations of Selected Elements," which describes this, 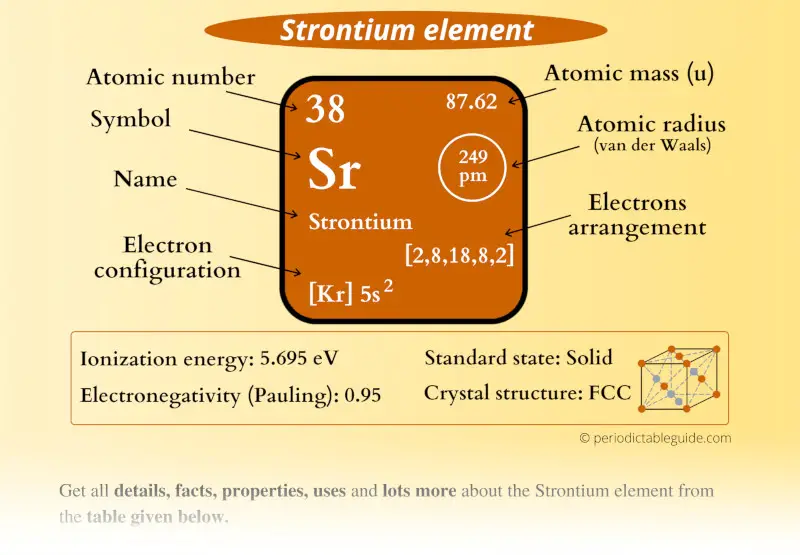
It should not be surprising, but isotopic abundances (% of each isotope) can vary between samples. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
